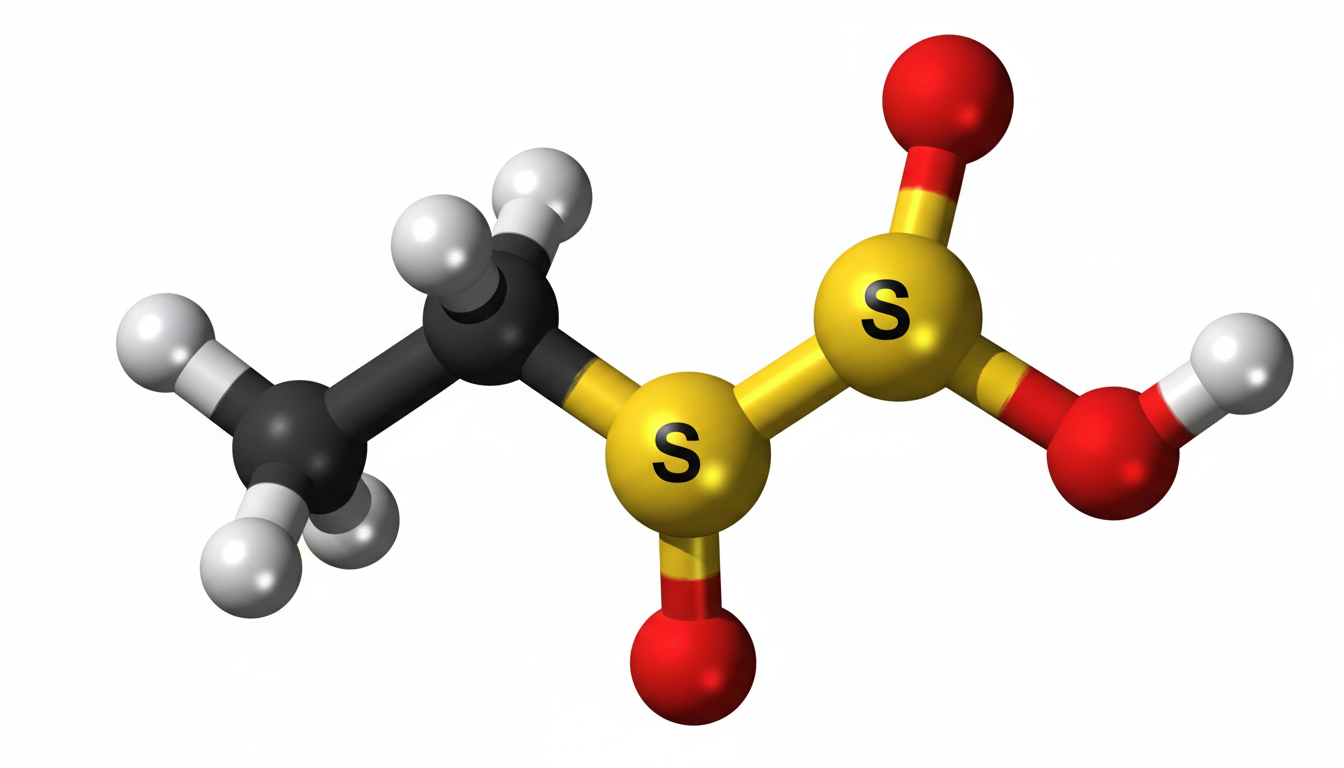
Ethylene sulfate is an important chemical compound that plays a role in various industrial and scientific applications. Although it is not as widely known as some other chemicals, it has unique properties that make it useful in specialized processes. Understanding its structure, uses, and safety considerations is essential for professionals working in chemistry-related fields.
What is Ethylene Sulfate?
Ethylene sulfate is an organic compound that belongs to the class of sulfate esters. It is derived from ethylene glycol and sulfuric acid. The compound typically appears as a colorless liquid and is known for its reactivity and stability under certain conditions. Due to its chemical nature, it is mainly used in controlled environments such as laboratories and industrial settings.
Chemical Properties
Ethylene sulfate has distinct chemical properties that make it suitable for specific reactions. It is relatively stable but can react with water and other nucleophiles. The compound has a cyclic structure, which contributes to its reactivity. It is also soluble in many organic solvents, making it versatile for chemical synthesis and experimentation.
Industrial Applications
In industry, ethylene sulfate is primarily used as an intermediate in chemical synthesis. It can be involved in the production of polymers, resins, and other complex organic compounds. Researchers also use it to study reaction mechanisms and develop new materials. Its ability to participate in ring-opening reactions makes it valuable in advanced chemical processes.
Laboratory Use
In laboratory environments, ethylene sulfate is used for research and experimental purposes. Scientists often utilize it to explore chemical reactions and to synthesize new compounds. It serves as a model compound in organic chemistry studies, helping researchers understand reaction pathways and molecular behavior.
Safety and Handling
Handling ethylene sulfate requires proper safety precautions. Like many chemical compounds, it can be hazardous if not used correctly. Direct contact with skin or eyes should be avoided, and it should only be handled in well-ventilated areas. Protective equipment such as gloves and goggles is recommended to minimize risks.
Environmental Impact
The environmental impact of ethylene sulfate depends on how it is managed and disposed of. Improper disposal can lead to contamination of water and soil. Therefore, industries and laboratories must follow strict guidelines to ensure safe handling and disposal. Responsible usage helps reduce potential harm to the environment.
Advantages and Limitations
One of the main advantages of ethylene sulfate is its reactivity, which makes it useful in various chemical processes. However, this same property can also be a limitation, as it requires careful handling. Its specialized applications mean that it is not commonly used outside of research and industrial contexts.
Conclusion
Ethylene sulfate is a valuable compound in the field of chemistry, offering unique properties that support research and industrial development. While it provides many benefits, it also requires careful handling due to potential risks. By understanding its characteristics and uses, professionals can utilize it effectively and safely in their work.